Cardiovascular implants and extracorporeal systems - Vascular prostheses - Tubular vascular grafts and vascular patches - ISO 7198
DIN EN ISO 7198 – Cardiovascular implants and extracorporeal systems – Vascular prostheses – tubular vascular grafts and vascular patches
This standard specifies requirements for the evaluation of vascular prostheses. It is applicable to sterile tubular vascular grafts implanted by direct visualization surgical techniques intended to replace, bypass or form shunts between segments of the vascular system. Vascular prostheses made of synthetic textile materials and synthetic non-textile materials are withing the scope as well as from non-viable biological origin. Compound, coated, composite and externally reinforced vascular prostheses are within the scope of this standard.
Test methods included for tubular vascular grafts or vascular patches are:
- Permeability
- Porosity (planimetric, gravimetric, microscopic)
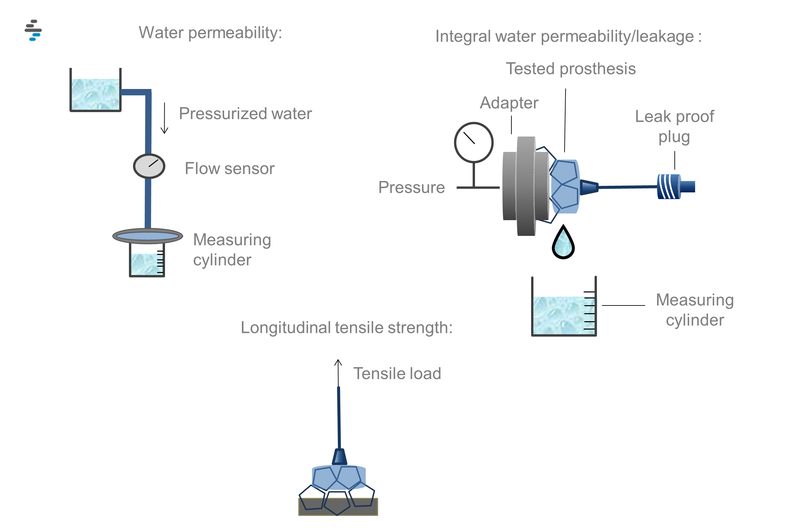
- Water permeability
- Integral water permeability/ leakage
- Water entry pressure
- Strength
- Pressurized burst strength
- Longitudinal tensile strength
- Circumferential tensile strength
- Diaphragm pressurizes burst strength
- Probe burst strength
- Strength after repeated puncture
- Length
- Relaxed internal diameter
- Pressurizes internal diameter
- Wall thickness
- Suture retention strength
- Kink diameter/ radius
- Dynamic radial compliance