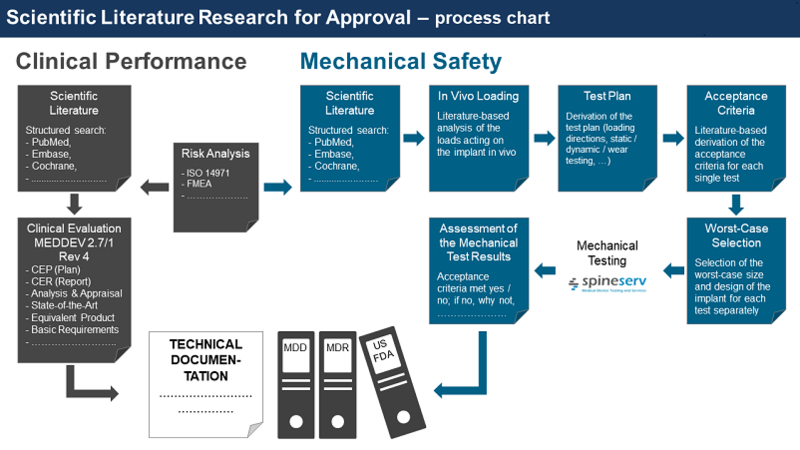
Scientific Literature Research
The requirements for product approval have increased worldwide in recent years. Scientifically sound literature research is a prerequisite for Clinical Evaluation, Biomechancial Documentation, and, thus, for the overall approval, as well as for research and development of medical devices (state of the art).
As a basic requirement for mechanical safety and performance as well as clinical evaluation, structured literature researches are an integral part of our scientific consulting portfolio.
We are characterized by a multidisciplinary background (engineering, mechanics, biomechanics, medicine, biology) and bring each discipline into the focus of implant and material testing. This is critical to the successful development of the most appropriate testing procedures for all types of implant applications. Our multidisciplinary expertise ensures an in-depth understanding of our results, taking into account both the technical and medical state of the art.